
The global medical affairs platform for efficiency, compliance, and impact across the pharmaceutical product life cycle, purpose-built to plan and manage evidence generation, grant programs, and communication activities.
Introducing iEnvision® 3.3
- EnvisionOne Insights integration
- doDOC presentation review integration
- Support for Adobe Sign
- Enhancements to external user password reset
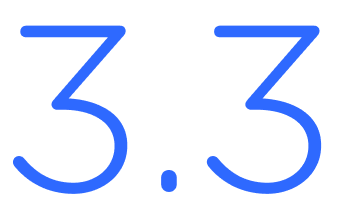

iEnvision Product Demo
Ready to learn how iEnvision can help your team drive smarter, faster outcomes? Request a demo today!
AI & Technology
(formerly Operational Excellence)
The Operational Excellence team have extensive experience across medical communications and medical affairs operations. They work closely with iEnvision’s teams to develop and implement customized strategies for integration of new processes/technology, adoption planning, and change management

Policy, process, and compliance
Our medical affairs experts understand what it takes to ensure successful development and implementation of governance and processes. We can support you in:
- Operational policy evaluation and development
- Implementation of industry best practices

Adoption planning and change management
Our experienced team will design a communication program providing alignment on impending changes, promoting benefits of the solution, and ensuring adoption across stakeholders.
- Program adoption
- Communication and training
- Evaluation and optimization

Business data and technology management
Stretched for resources or time? Our dedicated, skilled administrators help you realize the most from your technology tools by keeping accurate, up-to-date records for you.
- Medical affairs technology coordination
- Compliance monitoring
- Data analysis and metrics
Our dedicated in-house team have years of experience in the design, development, and implementation of software solutions, and are well placed to provide answers regarding:
- Platform and custom configurations
- Quality control
- Validation
- Hosting
- System integrations and APIs
Our Client Engagement team will help you get the most out of your solutions every day, providing:
- A dedicated point of contact
- Access to business experts for your programs of interest
- Guidance throughout iEnvision implementation
- Multiple training options for your team and third-party suppliers
- Hotline support for technical and training questions
- Ongoing support during maintenance and upgrades

Our holistic AI solution approach that aligns platform, people, and process to drive the medical insights journey with the highest performance
