Envision Pharma Group is a global, technology-enabled strategic partner to pharmaceutical, biotech, and medical technology companies, helping translate scientific and medical innovation into better patient outcomes.
We combine deep life sciences expertise, strategic insight, and AI-powered technology to help clients create, communicate, and convert value – delivering smarter, faster, measurable results.
With over 20 years of experience and trusted partnerships across 400+ life sciences companies, including all top 20 pharma, we deliver integrated, scalable solutions across the product lifecycle – from medical communications to commercialization, market access, revenue management, and compliance.
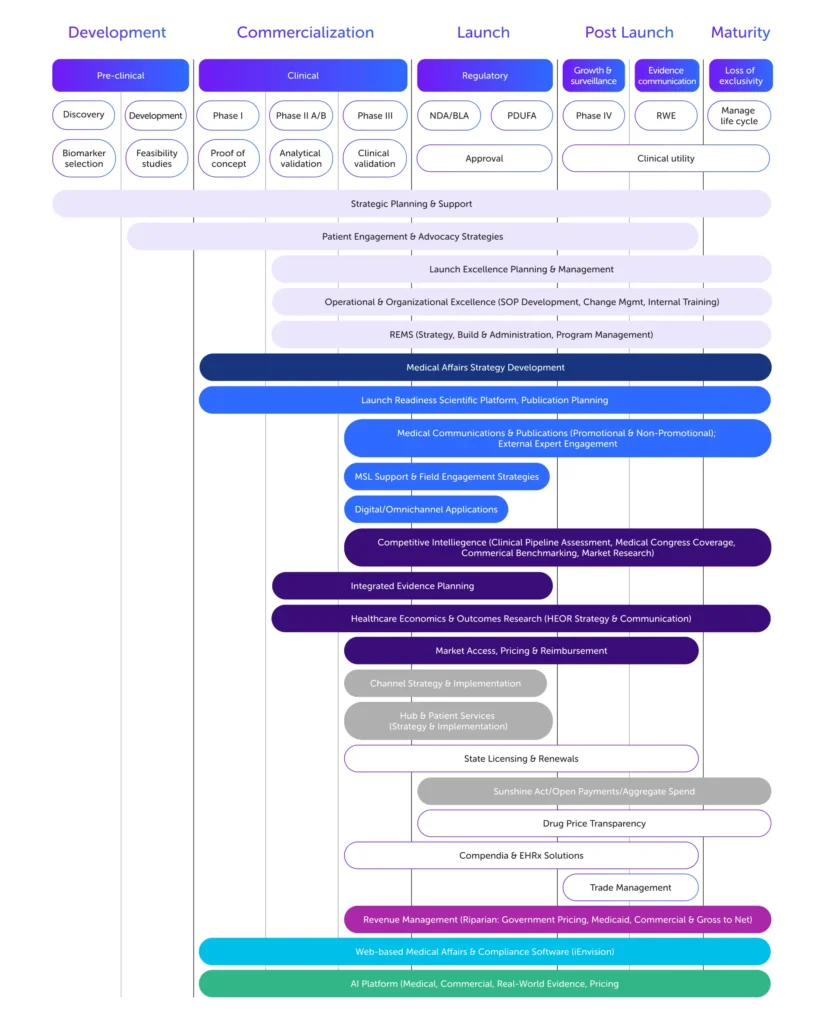
We start with your science, medical and commercial strategy, and stakeholders. Whether you’re preparing for launch, navigating market access, scaling, or transforming operations, our experts partner with you to connect data, insights, and execution, driving outcomes while reducing risk.



Our services aren’t siloed or one-size-fits-all. Whether you’re leveraging our technology, engaging with our consulting teams, or outsourcing operations through managed services, you benefit from the full depth of our expertise and proprietary tools.
Strategic service solutions – covering medical communications, strategic consulting, commercialization, and revenue management and market access – are powered by our AI-enabled technology platforms and optimized workflows.
Technology solutions – our technology comes with expert guidance and hands-on support.
Managed services solutions – built to scale, our managed services allow you to outsource functions while remaining grounded in the same insights we deliver through our strategic advisory.